Examination of the placenta is a procedure all midwives are familiar with, but why is it so important and what can the placenta reveal about maternal and neonatal wellbeing?
Performed as soon as possible after birth, examination of the placenta is a unique opportunity to explore and understand the intrauterine environment. It also provides a record of events that may be associated with adverse pregnancy outcomes (Ravishankar & Redline 2019).
The Normal Placenta
At term, a fresh, healthy placenta has the following characteristics:
- Approximately 15 to 20 cm (up to 22 cm) in diameter
- Approximately 2.0 to 2.5 cm thick
- Weighs about 500 to 600 g or 1/6 of the baby’s birth weight.
(PBB 2015; Leyto & Mare 2022)
These measurements may vary considerably depending on factors such as:
- The mode of birth
- Timing of cord clamping
- The time elapsed between delivery and examination.
(Akhavan et al. 2022)
Why Examine the Placenta?
Schollenberg et al. (2019) suggest that examination of the placenta is important because it reflects the pathophysiology of both the mother and the fetus. It can offer important diagnostic, prognostic and therapeutic information as well as potentially revealing information that might be relevant to future pregnancies. It may also provide vital clues regarding any potential causes of neonatal illness or death.
As Jaiman (2015) notes, the importance of the placenta is beyond doubt - it is an easily accessible source of information that reflects the intrauterine environment, providing insight into clinical problems that may not become evident for hours or even days after delivery, as well as being predictive of future pregnancies.
The Examination Technique
Examination of the placenta and membranes should take place as soon as possible following birth to ensure that they are complete and that no further actions are required before the parent is discharged or transferred to the ward.
After explaining the procedure to the parents, the placenta should be examined on a flat surface, in a place where there is adequate lighting, with the fetal surface facing up. If cord blood samples are required then a needle and syringe should also be readily available (Nurse Key 2019).
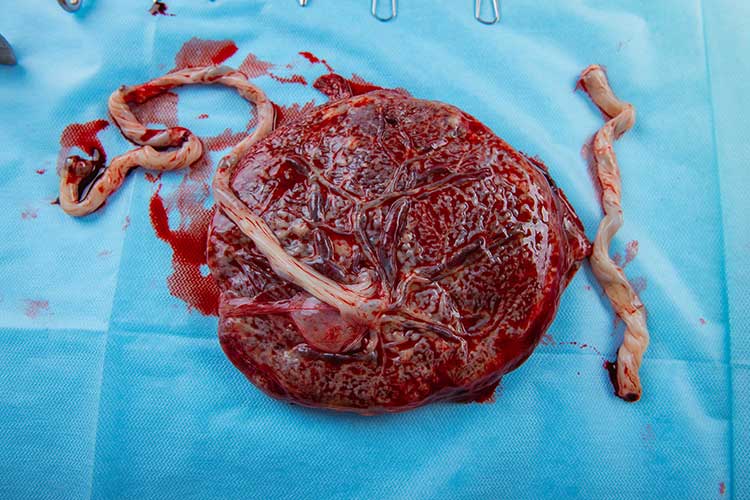
The Fetal Side
The fetal surface of the placenta should have a shiny, grey, translucent appearance so that the colour of the underlying maroon villous tissue of the maternal side can also be seen (PBB 2015).
Occasionally, irregularities such as succenturiate lobes are seen. These are smaller accessory placental lobes that are separate from the main disc of the placenta (Rathbun & Hildebrand 2022).
Insignificant changes may also occur, for example, infarctions due to the presence of fibrin deposits, or lime salt deposits that may make the surface feel gritty (Nurse Key 2019).
The Membranes
The membranes consist of two layers: the amnion and the chorion. These both need to be examined for insertion, colour, thickness and surface characteristics. By lifting the cord and holding the placenta up, both layers of membranes can be inspected for completeness. There should be a single hole present where the baby has passed through. The membranes should then be separated by pulling the amnion back over the base of the umbilical cord to ensure that both the amnion and chorion are present (Jaiman 2015).
The Umbilical Cord
The cord is usually inserted in the centre of the fetal surface with blood vessels branching outwards. At term, the typical umbilical cord length is 55 to 60 cm, with a diameter of 2.0 to 2.5 cm (Murphy 2022).
Jaiman (2015) notes that spiralling of the blood vessels should be present, most commonly with a left twist or in a counter-clockwise direction. This is important because cords with a significantly higher number of twists, and, perhaps those without twists, are associated with higher morbidity and mortality.
A normal cord has two arteries and one vein, which are covered by the amnion. The absence of one umbilical artery is a fairly common anomaly, occurring in approximately 1% of deliveries. Absence of an artery is more common with multiple pregnancies and velamentous cord insertions where the cord inserts on the edge of the placenta along the chorioamniotic membranes. Approximately 20% of infants who have a missing artery also have other major congenital anomalies that may involve any organ system, though this is most often associated with renal agenesis (Jaiman 2015).
Premature infants tend to have thicker umbilical cords than term babies, whereas thin cords are often associated with placental insufficiency. Other abnormalities include oedema of the cord, which can be seen in hydrops fetalis, and inflammation of the cord, which may manifest as a yellow discolouration, or micro-abscesses if infection with Candida is present. Long cords greater than 75 cm at term are usually associated with true knots and fetal entanglements, whereas short cords are known to occur in disorders with decreased fetal movement and are associated with problems in neurological development (Jaiman 2015).
The Maternal Side
The maternal surface of the placenta should be dark maroon in colour and consist of 15 to 20 cotyledons, which are divided by septa (Nurse Key 2019). Any abnormalities such as areas of infarction should be noted, and if infection is suspected, both the fetal and maternal sides should undergo a placental swab (The Royal Hospital for Women 2021).
A histological examination of the placenta may be indicated if there are abnormalities (The Royal Hospital for Women 2021).
Multiple Pregnancy

The placenta and membranes for multiple births are more complex and require careful assessment, as a variety of combinations are possible. For example:
- Monochorionic, monoamniotic twins are very rare. They have just one placenta, one pair of amnion and chorion, and two cords.
- Monochorionic, diamniotic twins have one placenta and one outer chorion, but the shiny fetal surface should have two cords, each inside its own amnion.
- Dichorionic, diamniotic twins always have two separate placental units, each with two layers of membranes just like a singleton. They may, however, be side by side, and appear to be joined at first glance.
(Raising Children Network 2022)
Conclusion
After completing a thorough examination of all parts of the placenta and taking cord blood samples if required, the findings should be documented and discussed with the parent. Any concerns about an incomplete placenta should be flagged both to the parent (who may experience an increase in blood loss or possible infection) and other members of the midwifery team who will be responsible for postnatal care.
Topics
References
- Akhavan et al. 2022, ‘Pathologic Examination of the Placenta and its Benefits in Treatment Plan or Follow-Up Of Patients: A Cross-Sectional Study’, European Journal of Medical Research, vol. 27, no. 113, viewed 24 August 2023, https://eurjmedres.biomedcentral.com/articles/10.1186/s40001-022-00743-7
- Benirschke K, Burton GJ & Baergen RN 2012, 'Examination of the Placenta', Pathology of the Human Placenta, Springer, Berlin, Heidelberg, viewed 23 August 2023, https://link.springer.com/chapter/10.1007/978-3-642-23941-0_1
- Jaiman, S 2015, 'Gross Examination of the Placenta and Its Importance in Evaluating an Unexplained Intrauterine Fetal Demise', Journal of Fetal Medicine, vol. 2, viewed 23 August 2023, https://link.springer.com/article/10.1007/s40556-015-0060-0
- Leyto, SM & Mare, KU 2022, ‘Association of Placental Parameters with Low Birth Weight Among Neonates Born in the Public Hospitals of Hadiya Zone, Southern Ethiopia: An Institution-Based Cross-Sectional Study’, International Journal of General Medicine, vol. 15, viewed 24 August 2023, https://www.dovepress.com/association-of-placental-parameters-with-low-birth-weight-among-neonat-peer-reviewed-fulltext-article-IJGM
- Murphy, A 2022, ‘Umbilical Cord’, Radiopaedia, 31 July, viewed 23 August 2023, https://radiopaedia.org/articles/umbilical-cord
- Nurse Key 2019, Examination of the Placenta and Membranes, Nurse Key, viewed 23 August 2023, https://nursekey.com/examination-of-the-placenta-and-membranes/
- Pregnancy Birth and Beyond 2015, Checking the Placenta, PBB, viewed 23 August 2023, https://www.pregnancy.com.au/checking-the-placenta/
- Raising Children Network 2022, Fraternal Twins, Identical Twins and Other Types of Twins, Raising Children Network, viewed 23 August 2023, https://raisingchildren.net.au/pregnancy/health-wellbeing/twin-pregnancy/twins
- Rathbun, KM & Hildebrand, JP 2022, ‘Placenta Abnormalities’, StatPearls, viewed 23 August 2023, https://www.ncbi.nlm.nih.gov/books/NBK459355/
- Ravishankar, S & Redline, RW 2019, 'The Placenta', Handbook of Clinical Neurology, vol. 162, viewed 23 August 2023, https://www.sciencedirect.com/science/article/pii/B9780444640291000035
- The Royal Hospital for Women 2021, Placental Examination and Indications for Referral to Pathology, New South Wales Government, viewed 24 August 2023, https://www.seslhd.health.nsw.gov.au/sites/default/files/documents/placentalexamreferpathology21.pdf
- Schollenberg E, Lee AF, Terry, J & Kinloch, M 2019, 'Placenta and Pregnancy-Related Diseases', Gynecologic and Obstetric Pathology, vol. 2, Springer, Singapore, viewed 23 August 2023, https://link.springer.com/chapter/10.1007/978-981-13-3019-3_15
 New
New 
