What is Epigenetics?
Published: 20 April 2015
Published: 20 April 2015

Epigenetics is the study of how the expression of DNA can be changed without changing the structure of DNA itself.
Understanding what causes disease is essential to not only create treatments but also to work towards ways of preventing the disease in the first place. While historically, the focus has been on the 'nature versus nurture' debate, relatively recent advances show that the argument may not be quite so black and white. So, let's review.
Each cell in our body has a nucleus. Inside each nucleus, there are 23 pairs of chromosomes. Each chromosome is made up of tightly packed DNA. Combinations of varying lengths of DNA make up genes. Genes are inherited from your parents and are the instructions that dictate the synthesis of proteins that make up new cells. Seems a lot to fit into each tiny nucleus!
In order to fit in the entire DNA required for each chromosome, it is coiled tightly around histones. A histone and DNA together is called a chromatin. Chromatins can be 'open' to allow reading of the genes or 'closed', depending on where the specific cell is located and its needs at the time (for example, chromatins controlling the synthesis for melatonin for skin pigmentation would be open in the skin cells but closed in the cells of the heart).
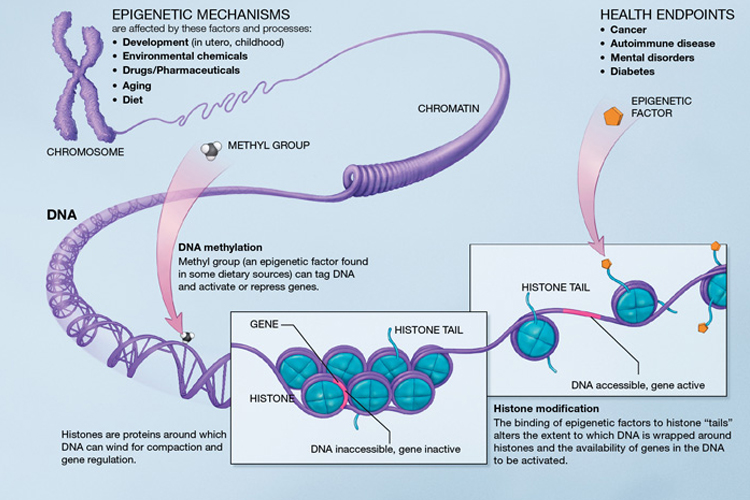
While genetics looks at the expression of the genetic code, epigenetics is the study of the other factors that influence how and when a gene is expressed. After much research in the area of epigenetics, the developmental origins of health and disease hypothesis was proposed, which contends that ‘during early life (at conception, and/or during fetal life, infancy and early childhood) the environment induces changes in development that have long term impact on later health and disease risk’ (International Society for Developmental Origins of Health and Disease).
It is believed that these changes in how an individual develops are caused by changes in the expression and/or the action of genes. Research involving professionals from many different specialties is identifying biomarkers (the alternations in gene expression that are correlated to a specific outcome) related to many disease processes such as cancer, autoimmune disease and 'lifestyle conditions' such as obesity.
Mice have a sequence of DNA that codes for the agouti gene. This gene dictates the fur colour of mice by working on the melatonin receptors at the hair follicle to influence the amount of pigment in the fur. The expression of this gene leads to dark fur. However, for a brief period during the growth of each hair, a change occurs in the level of pigmentation, leading to a yellow banding. This has the effect of creating varying degrees of grey-brown fur in normal mice.

However, in agouti variable yellow (Avy) mice, the period of yellow pigmentation during the growth of the hair is prolonged. This not only results in the mouse's fur colour appearing significantly more yellow but also causes the mouse to become obese, diabetic and prone to tumour development. These effects result from the fact that in Avy mice, the effect on melatonin occurs not only at the site of the hair follicle but also throughout the entire body. This demonstrates genetics. Let's now delve into the epigenetics of mice.
Within this group of Avy mice, all with genetic coding for changes throughout the hair follicles and the entire body, there are some that are genetically identical, have grey-brown fur and do not suffer from the systemic disease that plagues their siblings. They are known as pseudoagouti pups.
These lucky mice have an increased number of methyl groups that attach to the DNA sequence that encodes for the agouti gene (the defective gene that results in increased yellow pigmentation of hair, obesity, diabetes and increased tumour growth).
Methyl groups block the transcription of the DNA that they are attached to. The unfortunate mice with yellow fur have fewer methyl groups attached to the same DNA sequence, meaning there is nothing stopping the defective gene from being encoded to its full capacity.
In this case, the DNA is the same. The genes are the same. The difference exists in how the DNA is packaged.
Researchers then found they could increase the number of pseudoagouti pups born to an Avy mother by feeding her a diet fortified with folic acid and vitamin B 12, among other methyl-donors (substances that readily donate their extra methyl groups to DNA for use in methylation) during pregnancy. This affected not only the offspring but went on to benefit the offspring of the offspring!
Factors that reduce methylation in mice have also been found. These include prenatal exposure to bisphenol A (BPA - found in some plastics) and low levels of alcohol exposure.
Interestingly, it isn’t only the prenatal environment that affects methylation. Wearer et. al (2004) found that increased attention of a mother to her pups in the form of licking and grooming was linked with methylation in the glucocorticoid receptor in the hippocampus. These pups grew up to be calmer and less fearful in adulthood.

Mammals also make use of the methyl groups. Right now, scientists are working on ways to reduce exposure to substances that may reduce methylation and increase environmental exposure to factors that increase methylation (i.e. with maternal diet improvements) to reduce the effects of dysfunctional DNA sequences in humans. The implications of this are significant. Imagine having a way to avoid inherited conditions without changing DNA! That is the amazing, incredible area of epigenetics.
Could epigenetics put to rest the debate between nature and nurture?